Rajasthan Board RBSE Class 12 Chemistry Chapter 5 Surface Chemistry
RBSE Class 12 Chemistry Chapter 5 Test Book Type Questions
RBSE Class 12 Chemistry Chapter 5 Multiple Choice Questions
Question 1.
The equation for adsorption isotherm is :
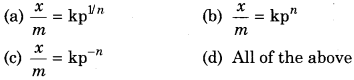
Question 2.
Shape selective catalysis is that reaction which is catalysed :
(a) by enzyme
(b) by zeolite
(c) by platinum
(d) by Zeigler-Natta Catalyst
Question 3.
The incorrect statement for physical adsorption is :
(a) adsorption is reversible at solid surface.
(b) the amount of adsorption increases on increasing the temperature.
(c) adsorption is spontaneous process.
(d) the enthalpy and entropy are negative.
Question 4.
Which of the following has minimum gold number?
(a) Gelatin
(b) Albumin of egg
(c) Gum arabic
(d) Starch
Question 5.
As2S3 colloid is negatively charged then in which of following its coagulation capacity will be maximum ?
(a) AlCl3
(b) Na3PO4
(c) CaCl2
(d) K2SO4
Question 6.
The activity of enzyme is maximum at:
(a) 300 K
(b) 310 K
(c) 320 K
(d) 330 K
Question 7.
Lyophilic sol is more stable than lyophobic sol, because :
(a) colloidal particles have positive charge.
(b) colloidal particles have no charge.
(c) colloidal particles have negative charge.
(d) strong electrostatic repulsive forces are present between negatively charged colloidal particles.
Question 8.
The adsorption capacity of adsorbate can be increased :
(a) by increasing surface area.
(b) by grinding.
(c) by making porous.
(d) all of the above
Question 9.
Which of the following is not a surface phenomenon ?
(a) Homogeneous catalysis.
(b) Mixing of solids.
(c) Formation of rust.
(d) Electrolytic process.
Question 10.
Arsenic sulphide sol is negatively charged. Which of the following has maximum capacity to convert it as
precipitate ?
(a) H2SO4
(b) Na3PO4
(c) CaCl2
(d) AlCl3
Question 11.
The method of blood purification in human body is :
(a) Electrophoresis
(b) Electric osmosis
(c) Dialysis
(d) Coagulation
Question 12.
The colloidal solution of red colour is obtained by adding some drops of dil HCl in precipitate of fresh ferric oxide. This process is called :
(a) Precipitation
(b) Dialysis
(c) Protection
(d) Dissociation
Question 13.
The zig-zig motion of colloidal particles is studied by:
(a) Zingmondy
(b) Ostwald
(c) Robert Brown
(d) Tyndall
Question 14.
Chromatography is based on:
(a) Physical adsorption
(b) Chemicaładsorption
(c) Hydrogen bond
(d) Adsorption
Question 15.
Gold number is related to :
(a) Electrophoresis
(b) Purple of Cassius
(c) Protective colloids
(d) Amount of pure gold
Answer:
1. (a) 2. (b) 3. (b) 4. (a) 5. (a) 6. (b) 7. (c) 8. (d) 9. (d) 10. (d) 11. (c) 12. (a) 13. (c) 14. (a) 15. (c)
RBSE Class 12 Chemistry Chapter 5 Very Short Answer Type Questions
Question 1.
Explain why colloidal particles present in colloidal solution are good adsorbent ?
Answer:
Colloidal particles have large surface area, therefore colloidal particles present in colloidal solution are good adsorbent.
Question 2.
Which type of colloid is cheese?
Answer:
Cheese is a gel in which dispersed phase is liquid and dispersion medium is solid.
Question 3.
Write one example of homogeneous and heterogeneous catalysis.
Answer:
Example of homogeneous catalysis :
2SO2(g) + O2(g) \(\underrightarrow { NO_{ (g) } } \) 2SO3(g)
Example of heterogeneous catalysis :
N2(g) + 3H(g) \(\underrightarrow { Fe_{ (s) } } \) 2NH3(g)
Question 4.
Colloidal solution exhibits Tyndall effect. Give two reasons.
Answer:
Colloidal solutions exhibit Tyndall effect due to two reasons which are as follow :
- Tyndall effect takes place due to scattering of light form the surface of colloidal particles.
- The scattering of light is due to differences of refractive index of dispersed phase and dispersion
medium..
Question 5.
Explain why alum is used to prevent bleeding from wound of body.
Answer:
Blood is colloidal solution of substances like albumin in water. It has negatively charged particles. The fresh solution of alum or ferric chloride is added on flowing blood which coagulates blood by Al3+ or Fe3+ ions and stop flow of blood.
Question 6.
What is meant by multimolecular colloid?
Answer:
On dissolution a large number of atoms or smaller molecules of a substance aggregate together to form
species having size in the colloidal range (1-1000 mm). These speices thus formed are called multimolecular colloids. e.g. A gold sol may contain particles of various sizes having many gold atoms.
Question 7.
Write two differences in adsorption and absorption :
Answer:
Differences between adsorption and absorption :
| Adsorption | Absorption |
|
1. It is the process in which a substance is adsorbed at the surface of another substanc |
1. It is process in which a substance is uniformly distributed throughout the bulk of another substance. |
| 2. it is surface process which takes place at surface only. | 2. It is a bulk process because it takes place throughout the substance. |
Question 8.
Which adsorbent is used to remove hardness of water ?
Answer:
The group e.g. -COOH, -SO3H, -NH2 etc. present in organic polymers are capable of adsorbing specific ions from solution or water. They used to soften the hard water.
Question 9.
Define sorption.
Answer:
When the phenomenon of adsorption and absorption take place simultaneously then the process is called sorption.
Question 10.
Write a chemical reaction of auto catalyst.
Answer:
CH3COOC2H5 + H2O \(\underrightarrow { H^{ + } } \) CH3COOH + C2H5OH
In this reaction, CH3COOH acts as auto catalyst.
Question 11.
Which catalyst and promoter are used in Haber’s process ?
Answer:
Catalyst and promoter used in Haber’s process are Fe and Mo respectively.
Question 12.
On which phenomenon does enzyme catalysis work ? Which scientist proposed this phenomenon?
Answer:
Enzymes are complex nitrogeneous organic compounds produced by living cells which catalyze the biochemical reactions occurring living organism. Enzyme catalysis works on enzyme-substrate binding. This phenomenon is proposed by Michaelis and Menten.
Question 13.
Define gold number.
Answer:
The gold number is defined as the number of milligrams of a hydrophilic colloid that will just prevent the precipitation of 10 mL of a gold sol on the addition of 1 mL 10% NaCl solution.
Question 14.
Explain why finely divided substance is more effective adsorbent.
Answer:
Adsorption is a surface phenomenon. The extent of adsorption depends on the surface area, therefore adsorption is directly proportional to surface area. A finely divided substance like nickel, platinum and porous substances like charcoal, silica gel provide large surface area. Hence, finely divided substances behave as good adsorbent.
Question 15.
Write an example of each type of emulsion.
Answer:
- Oil-in-water (O/W): milk
- Water in oil (W/O): Butter
Question 16.
What is purple of cassius ?
Answer:
Purple of cassius is a purple pigment formed by the reaction of gold salts with tin (II) chloride. It has been used to impart red colouration as well as to determine the presence of gold as a chemical test.
Question 17.
Write the name of catalyst which convert methanol to gasoline.
Answer:
Zeolite catalyst ZSM-5 is used for converting methanol into gasoline.
Question 18.
Explain why soaps do not show saponification in acidic medium?
Answer:
Saponification value of soaps depends on the amount of base required to saponify therefore soaps do not show saponification in acidic medium.
Question 19.
Write an equation of induced catalysis.
Answer:
2Na2SO3 + O2 \(\underrightarrow { fast }\) Na2SO4
Na3AsO3 + O2 → No reaction
Na2SO3 + Na3AsO3 +O2 → Na2SO4 + Na3AsO4
Question 20.
Classify the following in lyophilic and lyophobic colloid :
(a) As2S3
(b) Gum
(c) Starch
(d) Au sol.
Answer:
Lyophilic colloids : Gum and Starch
Lyphobic colloids : As2S3 and Au sol.
RBSE Class 12 Chemistry Chapter 5 Short Answer Type Questions
Question 1.
Explain the mechanism of micelle formation.
Answer:
Micelles are formed by association of such molecules in which both lyophilic and lyophobic parts are present. Such molecules are called surface active molecules.
Example :
Hydrocarbon chain in soap, sodium stearate C17H35COONa is lyophobic or hydrophobic while -COONa is lyophilic or hydrophilic. The hydrocarbon tails are in the interior of the micelle and C00– ions on the surface. The grease stain absorbed in to the interior of the micelle which behaves like liquid hydrocarbon. As the strain is detached from the fabric, the dirt particles sticking to the strain are also removed.
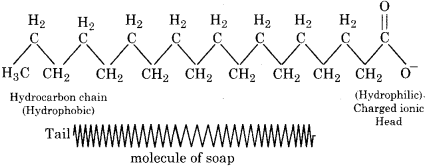
The formation of micelle and action of detergent can be explained by following figures.
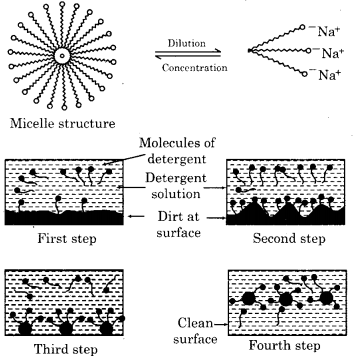
Question 2.
Write short notes on the following
(a) Dialysis
(b) Cottrell precipitator
Answer:
(a) Dialysis: The process of dialysis is based on the fact that colloidal particles can not be diffused from parchment membrane or cell membrane and cell wall while the ions of electrolyte are diffused.
The colloidal solution taken in a bag made of cellophane or parchment. The bag is suspended in water. The impurities of electrolyte are diffused slowly towards outside and pure colloidal solution remains in dialysis apparatus. The process of the separating the paricles of colloids from those of crystalloids by means of diffusion through a suitable membrane is called dialysis.
(b) Cottrell precipitator: Smoke is a colloidal solution of carbon, arsenic compounds, dust etc in air, These carbon partieles are precipitated in direct contact with oppositely charged metallic plates for separation in smoke and the gases which evolved form chimney are smokeless. This precipitator is called cottrell smoke precipitator.
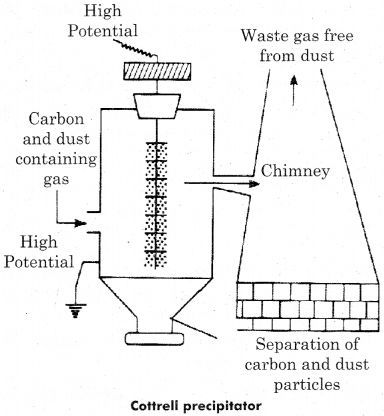
Question 3.
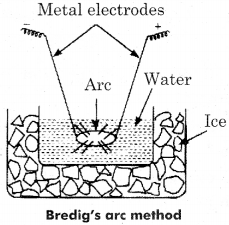
Describe the formation of colloidal solution of platinum in water dispersion method.
Answer:
Colloidal solutions of metals are obtained by this method. The metal whose sol is to be prepared is made as two electrodes immersed in dispersion medium such as water. The dispersion medium is kept cool by surrounding it with a freezing mixture.
An electric arc is struck between the electrodes the tremendous heat generated by the arc vaporises the metals which are condensed immediately in the liquid to give colloidal solution. The colloidal solution of Pt is prepared by this method. Such obtained colloids are water repellent so small quantity of KOH is mixed for their stability.
Question 4.
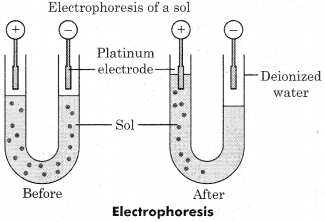
Discuss the process of electrophoresis with neat and labelled diagram.
Answer:
Electrophoresis:
The movement of colloidal particles towards oppositely charged electrodes in presence of electric filed is called electrophoresis. The movement of colloidal particles towards cathode is called positive electrophoresis and towards anode is called negative electrophoresis.
The nature of charge present on colloidal particle can be determined with the help of electrophoresis. Example:
Colloidal particles of As2S3 move towards anode in the presence of electric field so these colloidal particles are negatively charged.
Charged colloidal particles loose their charge by reaching oppositely charged electrode on passing electric current.
Now, neutral colloidal particles on aggregation, coagulate to form large particles.
Question 5.
Write a mathematical equation of Freundlich adsorption isotherm.
Answer:
Freundlich Adsorption Isotherm:
The variation of extent of adsorption (x/ m) with pressure (P) at a particular temperature was given mathematically by Freundlich in 1909. From the adsorption isotherm, the following observations can be easily made :
(i) At low pressure:
The graph is almost straight line which indicates that (x/ m) is directly proportional to the pressure. This may be expressed as:
\(\frac { x }{ m } \propto P\quad or\quad \frac { x }{ m } =kp\) … (1)
Where k is a constant.
(ii) At high pressure:
The graph becomes almost horizontal which means that x/ m becomes independent of pressure. This may be expressed by
\(\frac { x }{ m } =constant\quad \)
or
\(\frac { x }{ m } \propto P^{ o }\) ( ∵ Po = 1)
(iii) At medium pressure:
The value of + depends on the power of pressure whose value is between zero to one.
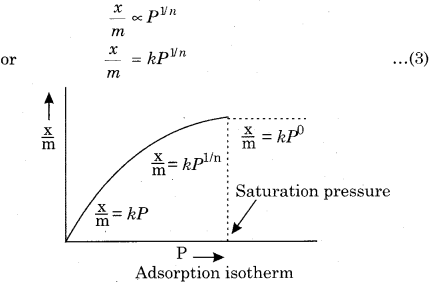
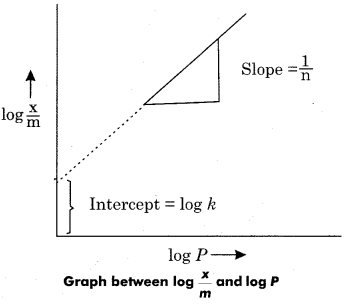
n and k are constant whose value depend on the nature of adsorbate and adsorbent. This relationship was first given by Freundlich. So it is also called Freundlich adsorption isotherm. By taking logarithm of both sides of equation (3).
![]()
Comparing eq. (4) to y’= c + mx so if graph is plotted between ![]() and log P, then a straight line is obtained.
and log P, then a straight line is obtained.
The slope of the line will be equal to \(\frac { 1 }{ n } \) and intercept will be equal to log k.
Question 6.
Write four differences between physical adsorption and chemical adsorption.
Answer:
Four differences between physical adsorption and chemical adsorption :
| Physical adsorption | Chemical adsorption |
| 1. The forces between the adsorbate molecules and the adsorbent are weak van der Waal’s forces. | 1. The forces between the adsorbate molecules and the adsorbent are strong chemical forces similar to chemical bonding. |
| 2. Low enthalpy of adsorption of the order of 2O to 4O kJ mol-1 | 2. High enthalpy of adsorption of the order of 80 to 240kJ mol-1. |
| 3. Usually occurs at low temperature and decreases with increase in temperature. | 3. It occurs at high temperature and increases with the increase in temperature. |
| 4. It is reversible in ture. | 4. It is irreversible. |
Question 7.
What is the difference between multimolecular colloid and macro-molecular colloid ?
Write an example of each.
Answer:
Differences between multimolecular and macro-molecular colloids :
table
| Multimolecular Colloids | Macromolecular Colloids |
| 1. They consist of aggregates of atoms or molecules which generally have diameter |
1. They consist of large size molecules (generally polymers) like rubber, nylon etc. |
| 2. The atoms or molecules are held by weak vander Waal’s forces. | 2. The molecules are flexible and can take any shape. |
| 3. Their molecular masses are not very high. | 3. They have high molecular masses. |
| 4. They have usually lyophobic character. Example: Sols of gold, sulphur etc. |
4. They have usually lyophilic character Example: Starch, proteins etc. |
Question 8.
What will be the observation in following conditions ?
(a) When light rays travel through colloidal solution ?
(b) Electric current is passed through colloidal solution ?
Answer:
(a) When light rays travel through colloidal solution:
When light rays are allowed to pass through a true solution then this light does not seen in solution till the eyes have not close in the path of direction of light. But when light beam is passed through colloidal solution then this light is seen in the form of bright line. The phenomenon is called Tyndall effect.
(b) Electric current is passed through colloidal solution:
Electric current is passed through colloidal solution then the movement of colloidal particles observe towards oppositely charged electrodes in presence of electric field. This phenomenon is called electrophoresis.
Question 9.
Give the characteristics of enzyme catalysis.
Answer:
Characteristics of enzyme catalysis:
- Highly effective:
Enzyme catalysts increase the rate of reactions by 108 to 1020 times as compared to the uncatalysed reaction. - Temperature and pH:
The enzyme catalysis is active at average temperature (37°C or 25-37°C) and certain pH (pH = 7) - Small amount:
Very small amount of enzyme increases the rates of reactions by 10% to 106 times. - Specificity:
Enzymes are very specific in nature. One enzyme can act as catalyst in reaction.
RBSE Class 12 Chemistry Chapter 5 Long Answer Type Questions
Question 1.
Explain the following with the help of diagram
(i) Tyndall effect
(ii) Brownian movement.
Answer:
(i) Tyndall effect:
When light beam is allowed to pass through a true solution in dark then light does not seen in solution till the eyes have not close in the path of direction of light but when light beam is passed through colloidal solution then this light is seen in the form of bright line. This phenomenon is called Tyndall effect.
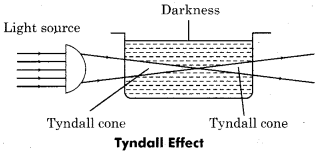
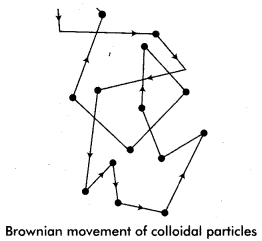
(ii) Brownian movement:
British Botanist Robert Brown (1828) found that the pollen grains present in water move in a continuous zig-zag movement, this movement is called Brownian movement.
Question 2.
Describe the following methods for the formation of colloidal solution
(i) Bredig-Arc method
(ii) Colloidal mill.
Answer:
(i) Bredig-Arc method:
Colloidal solutions of metals are obtained by this method. The metal, whose sol is to be prepared, is made as two electrodes immersed in dispersion medium such as water. The dispersion medium is kept cool by surrounding it with a freezing mixture.
An electric arc is struck between the electrodes. The tremendous heat generated by the arc vaporises the metals which are condensed immediately in the liquid to give colloidal solution. The colloidal solution of the metals are water repellant so small quantity of KOH is mixed for their stability.
(ii) Colloidal mill:
A colloidal mill is a machine that is used to reduce the particle size of a solid in suspension in a liquid, or to reduce the droplet size of a liquid suspended in another liquid. It consists of two metallic discs nearly touching each other and rotating in opposite directions to very high speed.
The space between the discs of the mill is so adjusted that coarse suspension is subjected to great shearing force giving rise to particles of colloidal size. Colloidal solutions of black ink, paints, varnishes, dyes etc are obtained by this method.
Question 3.
Write a note on shape selective catalyst Zeolite.
Answer:
Zeolite catalyst or shape selective catalyst:
Zeolites are small porous alumina silicates. Their general formula is M3 [(Al2O3)x (SiO2)y]3 mH<sub.2O where n is the oxidation state of metal (M).
Example:
Zeolite catalyst ZSM-5 is used in the conversion of alcohol into petrol (gasoline). The dehydration of alcohol takes place on pores of zeolite and forms a mixture of hydrocarbons. Also refer to article No. 5.7.4 (c).
Question 4.
Give the reasons
(a) Alum purifies drinking water.
(b) A substance can be both colloidal and crystalloid
(c) Sky appears blue.
Answer:
(a) Impure water contains soil particles, bacteria etc. These particles are negatively charged. Alum is added in water to remove them. Al3+ ions present in alum precipitates impurities present in water. Pure water is separated by decantation.
(b) Colloids:
These are not diffused from animal membrane.
Crystalloids:
These are diffused from animal membrane. But the classification of substance given by Grahm was not satisfactory completely, because a specific compound behaves as crystalloids in one solvent and also as colloid in other solvent.
Example:
The aqueous solution of NaCl is crystalloid while it behaves as colloid in benzene.
(c) The wavelength of blue colour is very less hence its scattering is very high. Due to this reason, sky appears as blue.
Question 5.
Discuss the factors affecting adsorption of gases on solid surface.
Answer:
The exact amount of a gas adsorbed at the surface of a solid depends upon the following factors:
(i) Nature of gas (adsorbate):
If critical temperature of gas is higher, the van der Waal’s forces of attraction will be greater so adsorption will be greater. Critical temperatures of a few gases are given below:
| Gas | H2 | N2 | CO | CH4 | CO2 |
| Critical Temperature K | 33 | 126 | 134 | 190 | 304 |
(ii) Nature of solid (adsorbent):
Greater the surface area per unit mass of the solid, greater is the extent of adsorption.
(iii) Effect of temperature:
Adsorption is accompained by evolution of heat.
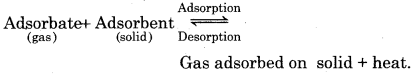
∆H = -ve. According to Le-Chateliers’s principle, the rate of adsorption decreases with increase in temperature.
(iv) Effect of pressure:
At constant temperature, adsorption increases with pressure. If the temperature is kept constant at low value, the effect will be large.